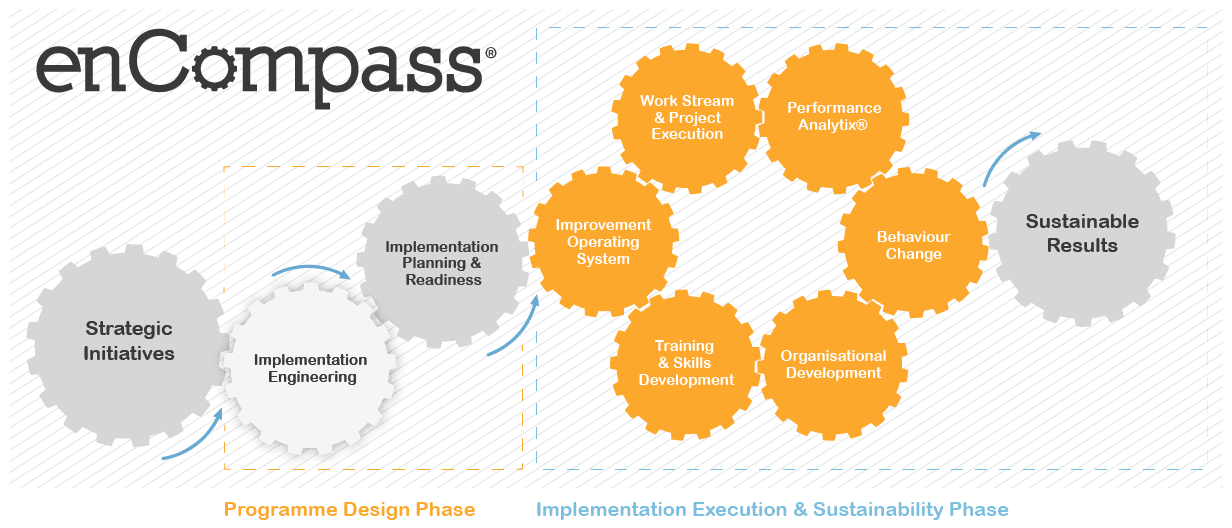
What is Implementation Engineers?
Ever wish that you could resolve a manufacturing problem easily while making your shareholders happy? Or use an outside firm and not feel buyers’ remorse? Wouldn’t it be wonderful if that outside firm found the cash to pay for the solution to your manufacturing issue that would make your shareholders